A Physician’s Summary
Andersen-Tawil Syndrome (ATS) is a genetic disorder which causes a distinctive pattern of features;
1) Episodes of muscle weakness and/or flaccid paralysis (i.e periodic paralysis)
2) Certain kinds of heart rhythm disturbances and
3) Differences in facial and body structure.
Clinical signs: Most ATS patients begin to have symptoms by the time they are 20, but later onset is not unusual. Heart palpitations and/or fainting are common signs, along with weakness that occurs on waking or following rest after exertion. Mild background weakness is common. ATS can cause learning disorders, and problems in such areas as planning ahead, abstract thinking, judging whether a behaviour is appropriate and screening out sensory input. This can resemble the behaviour of a child with ADD.
Potassium Levels: During attacks of ATS a patient’s potassium level may be within normal range, below normal or higher than normal. The potassium level may vary from attack to attack, with high potassium during one attack and low during the next one. Attacks may be triggered by low (or high) potassium, taking potassium supplement (in some patients), exercise, rest after exercise, sleep or other factors which vary from patient.
Heart Arrhythmias: ATS patients have a variety of heart rhythm disturbances. These may include a prolonged QT interval. The QT interval is a measurement of the time between the start of the Q wave and the end of the T wave in the heart’s electrical cycle. Normal QTc interval is 0.39 sec for men and 0.44 sec for women. (The small “c” after QT means the measurement has been adjusted to reflect the heart rate when the EKG was taken.) The length of the QT interval may vary with exercise, emotion or potassium level. Skeletal Affects: ATS patients often have facial and skeletal differences such as low-set ears, widely set eyes, a small jaw and chin, curved or crooked fingers (especially an inward curvature of the fifth finger as seen in the photo), webbing between the second and third fingers and/or toes (see photos), cleft palate, and curvature of the spine. Most ATS patients are small in stature, though this is not always the case.
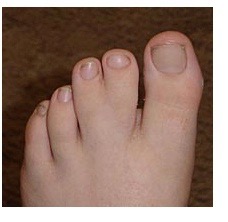
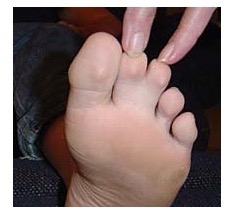
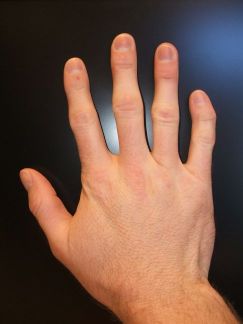
Family Assessment: When assessing a person for ATS, the doctor will look at all close relatives; parents, grandparents, brothers and sisters, children, even cousins, because the signs of the disorder can be spread out among family members. One might have paralytic attacks, another might have heart problems and the physical signs, like widely spaced eyes or webbing between toes, and have no weakness. The physical signs might be very subtle and easily overlooked by anyone other than an experienced clinician.
Screening: Every patient who has been diagnosed with periodic paralysis should be screened for ATS, with special emphasis on EKG screening for a prolonged QTc interval. Patients who have any symptoms or physical signs suggestive of ATS should be referred to a medical team experienced in ATS. An exercise EKG will usually be used to help make the diagnosis. The goal of the exercise test is to allow the individual to exercise for 10 or more minutes without reaching a heart rate in excess of 150-160 beats per minute while hooked up to a heart monitor. This exercise often “uncovers” a long QT interval which doesn’t show up when the patient is at rest.
Genetics and Inheritance: All identified ATS mutations have been found in the potassium channels KCNJ2 (Kir2.1), which is on chromosome 17, and KCNJ5 on chromosome 11. ATS is passed in an autosomal dominant pattern, that is, each child of a parent with ATS has a 50% chance of inheriting the mutation. This happens at the moment of conception. Genetic testing can identify mutations in only about 40% of clinically diagnosed ATS patients, as many mutations have yet to be identified.
Treatment: Treatment must be worked out individually for each patient, and may require adjusting from time to time. Treatment for ATS remains guided by symptoms and is sometimes frustrated by unusual responses of the heart and muscles to changes in potassium levels, unpredictable responses to medications, and an overall resistance to the medications used to treat heart rhythm disturbances.
Taking potassium improves weakness in some patients. In some families, taking oral potassium reduces heart rhythm problems but makes muscle weakness worse. In addition to the medications used for the periodic paralyses, (i.e. acetazolamide, dichlorphenamide, aldactone, dyrenium, etc) beta-blockers (esp. Atenolol) have been used successfully in some patients. Patients who have had a cardiac arrest or who continue to faint in spite of medication are sometimes given an implantable defibrillator or pacemaker, but this is rarely required. Patients must avoid hypokalemia and drugs that cause lengthening of the QT interval.
Drugs Which Patients With Prolonged QT Interval Should Avoid:
Note: Use this list as a starting point only. Clinicians should consult a reliable pharmacology source for authoritative information. The most easily accessible and reliable source is found at www.crediblemeds.org. Credible Meds has also posted a list of non-medication risks of prolonging the ECG QT interval. It is very useful as they have quantified the risk and posted links to supporting publications.
BETA-AGONISTS: Epinephrine a.k.a. adrenaline (many trade names) (found in local anaesthetics and asthma medications)
ANTIHISTAMINES: Seldane, Hismanal, Benadryl, Terfenadine, Astemizole, Diphenhydramine
ANTIBIOTICS: E-Mycin, EES, Erypeds, PCE, etc., Bactrim, Septra, Erythromycins, Trimethoprim, Sulfamethoxazole, Pentamidine, Pentam
IV HEART MEDICATIONS: Amiodarone HCI, Quinidine, Quinidex, Duraquin, Quiniqlute, Pronestyl, Norpace, Betapace, Lorelco, Vascor, Procainamide, Disopyramide, Sotalol, Probucol, Bepridil
GASTROINTESTINAL: Propulsid, Cisapride
ANTIFUNGALS: Nizoral, Diflucan, Sporanox, Ketoconazole, Fluconazole, Itraconazole
PSYCHOTROPICS: Norpramine, Viractil, Compazine, Stelazine, Thorazine, Mellaril, Etrafon, Trilafon, Haldol, Risperdal, ORAP, Amitriptyline, tricyclics, phenothiazine derivatives, Haloperidol, Risperidone, Pimozide
DIURETICS: Lozol, Indapamide. Many diuretics cause potassium wasting, and hypokalemia lengthens the QT interval.
References:
Andersen-Tawil Syndrome; National Library of Medicine: Aravindhan Veerapandiyan, MBBS, Jeffrey M Statland, MD, and Rabi Tawil, MD. (Strongly recommended)
1. Andersen, ED; et al: Intermittent muscular weakness, extrasystoles, and multiple developmental anomalies: a new syndrome? Acta Paediat. Scand. 60: 559-564, 1971. PMID: 4106724
2. Bazett H. An analysis of the time-relations of electrocardiograms. Heart 7:353-370, 1920. PMID: 16813841
3. Canun, S; Perez, N; Beirana, LG: Andersen syndrome autosomal dominant in three generations. Am. J. Med. Genet. 85: 147-156, 1999. PMID: 10406668
4. Chalak, W; Taha, A; Araoya, M; Rifaat, M. Middle East Hospital, Beirut, Lebanon: Torsade de pointes. Report of 18 cases. J. Med. Liban. 41(2):62-8, 1993; disc. 68-9. PMID: 8057345
5. Gould, RJ; Steeg, CN; Eastwood, AB; Penn, AS; Rowland, LP; De Vivo, DC: Potential fatal cardiac dysrhythmia and hyperkalemic periodic paralysis. Neurology. 35:1208-1212, 1985. PMID: 4022357
6. Katz, JS; Wolf, GI; Iannaccone, S; Bryan, WW; Barohn, RJ: The Exercise Test in Andersen Syndrome. Arch. Neurol. 56:352-356, 1999. PMID: 10190827
7. Kramer, LD; Cole, JP; Messenger, JC; Ellestad, MH: Cardiac dysfunction in a patient with familial hypokalemic periodic paralysis. Chest. 75: 189-192, 1979. PMID: 421555
8. Levitt, LP; Rose, LI; Dawson, DM: Hypokalemic periodic paralysis with arrhythmia. New Eng. J. Med. 286: 253-254, 1972. PMID: 5007210
9. Lisak, RP; Lebeau, J; Tucker, SH; Rowland, LP: Hyperkalemic periodic paralysis with cardiac arrhythmia. Neurology. 20:386, 1970. PMID: 5535005
10. Neuromuscular Disease Center, Washington Univ School of Medicine, St. Louis, MO.
11. Sansone, V; Griggs, RC; Meola, G; Ptacek, LJ; Barohn, R; Iannaccone, S; Bryan, W; Baker, N; Janas, SJ; Scott, W; Ririe, D; Tawil, R: Andersen’s syndrome: a distinct periodic paralysis. Ann. Neurol. 42:305-312, 1997. PMID: 9307251
12. Stubbs, WA: Bidirectional ventricular tachycardia in familial hypokalaemic periodic paralysis. Proc. Roy. Soc. Med. 69: 223-224, 1976. PMID: 1265019
13. Tawil, R; Ptacek, LJ; Pavlakis, SG; De Vivo, DC; Penn, AS; Ozdemir, C; Griggs, RC: Andersen’s syndrome: potassium-sensitive periodic paralysis, ventricular ectopy, and dysmorphic features. Ann. Neurol. 35:326-330, 1994. PMID: 8080508
14. Yoshimura, T; Kaneuji, M; Okuno, T: Periodic paralysis with cardiac arrhythmia. Eur. J. Pediatr. 1 0:338-343, 1983. PMID: 6628456
15. Plaster, N.M.; Tawil, R. Tristani-Firouzi, M.; et. az; Mutations in Kiru.1 Cause the Developmental and Episodic Electric l Phentoypes in Andersen’s Syndrome. Cell Vol 105, 511-519, May 18, 2001. PMID: 11371347
16. Andelfinger G, Tapper AR, Welch RC; et. al; KCNJ2 Mutation Results in Andersen Syndrome with Sex-Specific Cardiac and Skeletal Muscle Phenotypes; Am J Hum Genet 2002 Sep;71(3):663-8 PMID: 12148092
17. Tristani-Firouzi, M.; Jensen, J. L.; Donaldson, M. R.; et. al. Functional and clinical characterization of KCNJ2 mutations associated with LQT7 (Andersen syndrome). J. Clin. Invest. 110: 381-388, 2002. PMID: 12163457
18. Lu, C.-W.; Lin, J.-H.; Rajawat, Y. S. et. al. Functional and clinical characterization of a mutation in KCNJ2 associated with Andersen-Tawil syndrome. J. Med. Genet. 43: 653-659, 2006. PMID: 16571646
19. Stunnenberg BC, Raaphorst J, Deenen JCW, Links TP, ; et. al. Prevalence and mutation spectrum of skeletal muscle channelopathies in the Netherlands. Neuromuscul Disord. 2018 May;28(5):402-407. Epub 2018 Mar 9. PMID: 29606556.
20. Hasegawa K, Ohno S, Kimura H, Itoh H, Makiyama T,; et. al. Mosaic KCNJ2 mutation in Andersen-Tawil syndrome: targeted deep sequencing is useful for the detection of mosaicism. Clin Genet. 2015 Mar;87(3):279-83. Epub 2014 Mar 6. PMID: 24635491.
21. Kostera-Pruszczyk A, Potulska-Chromik A, Pruszczyk P, Bieganowska K, ; et. al. Andersen-Tawil syndrome: report of 3 novel mutations and high risk of symptomatic cardiac involvement. Muscle Nerve. 2015 Feb;51(2):192-6. Epub 2014 Nov 19. PMID: 24861851.
22. Kukla P, Biernacka EK, Baranchuk A, Jastrzebski M,; et. al. Electrocardiogram in Andersen-Tawil syndrome. New electrocardiographic criteria for diagnosis of type-1 Andersen-Tawil syndrome. Curr Cardiol Rev. 2014 Aug;10(3):222-8. PMID: 24827800
reviewed and updated May 2022
